Researc
The DNA in the living cell is packed, together with the proteins attached to it, in a molecular complex called chromatin. Although it may seem that the chromatin serves only as a packaging of genetic data within the DNA, the way in which the DNA is packed considerably influences cellular systems. For example, DNA that is tightly packed may become inaccessible to biological machinery which reads the DNA, possibly leading to inactivation of genes encoded in that DNA sequence.
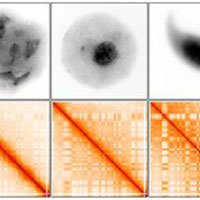
This current research examined the organization of the DNA during spermatogenesis – sperm development. Although spermatogenesis has been long studied, the way in which DNA is packed during this process has not been mapped in detail due to technological challenges. Now, researchers have met this challenge by using a novel technology, called Hi-C, which combines experimental molecular biology with computational analysis to measure the spatial organization of DNA.
Each day, millions of sperm cells are created in the human male body. One of the critical stages in the formation of sperm cells is meiosis (cell division). Early in meiosis, DNA is drastically reorganized as the chromosomes condense in preparation of the upcoming cell division. Furthermore, these condense chromosomes swap segments of DNA and in this way increase genetic variation.
The Israeli-American research team successfully isolated mouse sperm cells at the start of meiosis when the chromosomes are condensed and then used Hi-C to measure the spatial organization of the DNA. The researchers discovered that the spatial structure of the chromatin gradually strengthens during spermatogenesis, until it reaches its ultimate strength in the mature sperm. They suggest that this organization enables the sperm cells to activate a wide variety of genes during meiosis, enabling the cells to later gain the unique ability of producing all cell types after fertilization. According to Dr. Kaplan, “In the future, we intend to use this approach in order to understand how the genome’s spatial structure may influence fertility.”
This research was funded by the National Institutes of Health (NIH), Azrieli Foundation and the Henry and Marilyn Taub Scholarship.
Prof. Kaplan joined the Technion Rappaport Faculty of Medicine in 2016 and established an interdisciplinary laboratory for studying the spatial structure and function of genomes in health and disease.
Haia Khoury completed her BSc at the Technion’s Faculty of Biology and is currently pursuing an MSc in Biomedical Sciences at the Technion Rappaport Faculty of Medicine.
Click here to read the paper in Nature Structural & Molecular Biology