A Technion-led international research group deciphered the process of formation of hard and durable skeletons in the bodies of marine organisms

How do marine organisms produce hard tissues from the materials available to them, and under the hostile conditions that prevail under the waves? That question is the basis of a study by an international group led by Professor Boaz Pokroy, doctoral student Nuphar Bianco-Stein (as part of her Ph.D. thesis), and researcher Dr. Alex Kartsman from the Technion Faculty of Materials Science and Engineering, with the assistance of Dr. Catherine Dejoie from the European Synchrotron Radiation Facility (ESRF) in Grenoble, France.
The study, published in PNAS, focused on the involvement of magnesium-containing calcite in the aforementioned processes. Magnesium is a strong and light metallic element that plays many roles in the animal world, including in the human body. Calcite is a very common mineral that constitutes about 4% of the mass of the Earth’s crust. This material is an essential component in biomineralization, the process by which organisms form various structures – pearls, bones, shells, etc. – from the materials available to them.

“Biomineralization processes,” explained Prof. Pokroy, “build structures that surpass artificial products of engineering processes in many aspects, such as strength and resistance to fractures. So there is no doubt that we have a lot to learn from these biological processes, and that our findings may lead to improved engineering processes in a variety of areas.”
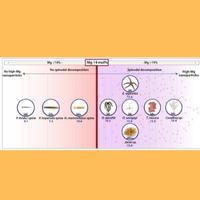
In the biomineralization process, organisms use various strategies to produce strong structures such as an external skeleton. The team from the Technion and ESRF showed in their study that one of the common strategies in this regard is the sedimentation of magnesium-rich nanoscale calcite particles within a magnesium deficient substance. According to Prof. Pokroy, “We have discovered that this phenomenon occurs in a huge variety of creatures, even creatures from different kingdoms in the animal world, and we estimate that it is even broader than what we have discovered. Therefore, it is likely to be a very general phenomenon.”
In the current study, the researchers focused on nine different organisms belonging to different kingdoms and phyla including brittle stars, red algae, starfish, coral, and sea urchins. The two main players in the process are, as mentioned, magnesium and calcite. The researchers found that the sedimentation of the calcite particles in the magnesium-poor substance creates compression efforts in the skeletons that increase their rigidity – without the need for mechanical compression used in the production of similar materials in classical engineering processes.
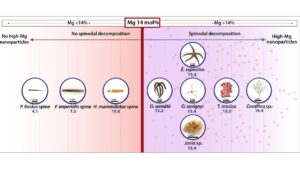
In brittle stars, the unique crystallization process takes place in the calcite lenses scattered on its arms – a kind of primitive but effective set of eyes for its needs. These lenses, which are similar in nature to tempered glass, focus sunlight on nerve centers that transmit information to the rest of the body through the nervous system. In contrast to tempered glass that man produces at high heat and under pressure, the brittle star lenses are created at the given water temperature and without external mechanical pressure other than the water pressure. An important step in this process is the transition of the calcite from an amorphous (disordered) phase – to the ordered crystalline phase.
The red algae that the international research team studied are the common algae found in shallow water, where they are subjected to external pressures of the sea waves and therefore must be resistant to tearing and fracture. Therefore, their cells are coated with the same strong nanoscale crystals of magnesium-calcite. These crystals form hollow micro-structures that increase their strength and durability.
Although brittle stars and red algae are very different creatures, they exhibit common structural aspects including the crystallization of strong structures in the process of sedimentation of magnesium-rich nanometer crystals in a substance characterized by low magnesium levels.
The researchers found that this crystallization process improves both the hardness of the material and the resistance to fractures. Moreover, they show in the study that even a slight reduction of the magnesium content in the substance doubles the hardness of the material by some 100%.
The study was supported by an EU grant from the ERC and was conducted in collaboration with the European Synchrotron Radiation Facility in Grenoble, France and the Argonne National Laboratory in Illinois, USA.